top of page
RESEARCH
The Bouhaddou Lab dissects biochemical signaling circuits, attempting to understand how signaling network connectivity and dynamics impact cell fate outcomes. Cellular signaling can be envisioned to be the "mind" of the cell, generating the thoughts (i.e. signaling) that eventually turn into actions (i.e. cellular phenotypes).
The lab is 50% “wet” and 50% “dry”, cultivating an interactive exchange between experimental and computational workflows. Experimentally, we focus on global mass spectrometry proteomics/ phosphoproteomics, cell culture-based virology, genetic and pharmacological perturbations, and molecular biology. Computationally, we specialize in big data bioinformatics, network modeling, and ordinary differential equation modeling.
We do not focus on one biological or disease area, instead our focus is on how post-translational modifications (PTMs), particularly phosphorylation, and protein-protein interactions (PPIs) impact biological phenotypes.
TECHNOLOGY
The Bouhaddou Lab does 50% "wet" and 50% "dry" science.


MOLECULAR BIOLOGY
MASS SPECTROMETRY PROTEOMICS
COMPUTATIONAL BIOLOGY
RESEARCH PROJECTS

Viruses as Biosensors
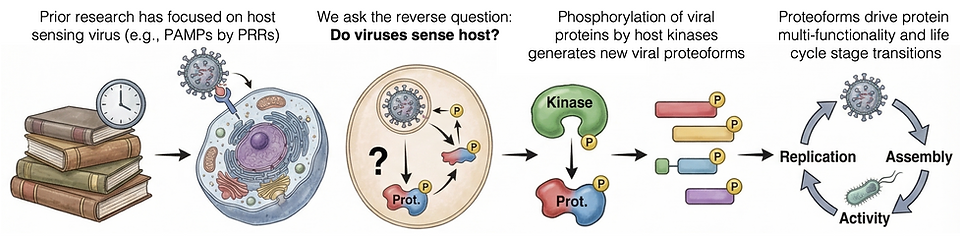
Decades of prior research has focused on how the host senses and responds to viruses, such as by the detection of pathogen-associated molecular patterns by host pattern recognition receptors.
Here, we study the reverse question:
Do viruses sense and respond to the host and, if so, what are the underlying biochemical mechanisms?
We study how phosphorylation of viral proteins by host kinases generates proteoforms that drive multi-functionality in response to host signaling states. We observe widespread, high-confidence human kinase motifs across viral families. Using a virus-agnostic framework, we define how post-translational modifications regulate virus-host protein-protein interactions and viral life cycle progression, and test whether viruses function as biosensors of host signaling states enabling real-time adaptation. We employ a systems biology approach integrating mass spectrometry phosphoproteomics, affinity purification–mass spectrometry (AP-MS), molecular virology, and bioinformatics.
Current Projects
Global Landscape of Human Kinase Motifs in Viral Proteomes. We used a hybrid computational and experimental approach to interrogate human kinase motifs across viral proteomes, integrating AlphaFold structural modeling with mass spectrometry phosphoproteomics to define the kinase pathways that phosphorylate viral proteins.
People: Kareem Alba, Declan Winters
Collaborators: Melody Li (UCLA), Oliver Fregoso (UCSC), and Ting-Ting Wu (UCLA)
KSHV LANA Protein. We study how phosphorylation of the Kaposi’s sarcoma–associated herpesvirus (KSHV) latency-associated nuclear antigen (LANA) by host DNA replication, stress, and cell cycle kinases regulates latency and episomal persistence across the cell cycle, assessing whether LANA functions as a biosensor of host signaling states, with extension to Epstein-Barr virus (EBV).
People: Sara Makanani (HHMI Gilliam Scholar), Angelina Park
Collaborators: Ting-Ting Wu (UCLA)
SARS-CoV-2 Membrane Protein. We study how phosphorylation of the C-terminal tail of SARS-CoV-2 membrane (M) protein impacts viral assembly and M protein multi-functionality.
People: Yennifer Delgado (NSF GRFP Scholar), Andrea Chan
Collaborators: Taha Taha (Gladstone), Melanie Ott (Gladstone)
Alphavirus Non-Structural Proteins. We study how host stress kinase-mediated phosphorylation of alphaviruses, including Sindbis virus (SINV) and Chikungunya virus (CHIKV), regulates replication complex function, viral protein production, and persistence, leading to debilitating arthralgia.
People: Declan Winters (T32-CMB Scholar), Justin Selby, Evan Wynn, Valerie Vandersall
Collaborators: Melody Li (UCLA)
HIV-1 Associated Comorbidities

We currently study how HIV-1 contributes to peripheral neuropathy and cervical cancer. Although antiretroviral therapy (ART) has transformed HIV care by controlling viral transmission and preventing AIDS, it does not prevent cases of peripheral neuropathy, cervical disease, and other diseases. For example, >50% of people living with HIV develop distal symmetric polyneuropathy (HIV-DSP), characterized by debilitating pain. We investigate how HIV-1 Vpr drives the secretion of neurotoxic factors from macrophages that impact Schwann cells of the peripheral nervous system. In parallel, we study cervical cancer, where women co-infected with HPV and HIV are ~6-fold more likely to progress to advanced disease despite ART, suggesting additional drivers of viral synergy. We define factors secreted by HIV-infected immune cells and their effects on cervical epithelial cell responses.
Current Projects
Peripheral Neuropathy. We study how HIV-1-infected macrophages, with and without Vpr, regulate the secretion of factors that drive Schwann cell toxicity.
People: Sophie Blanc (Geffen Scholar), Anthony Truong
Collaborators: Oliver Fregoso (UCSC), Maureen Su (UCLA), Anthony Covarrubias (UCLA)
Cervical Cancer. We study how secretomes from HIV-1-infected primary T cells dysregulate HPV-infected cervical cells, activating PI3K-AKT signaling to accelerate cervical cancer progression.
People: Charles Olwal (UCLA)
Collaborators: Nevan Krogan (UCSF), Yaw Bediako (Yemaachi)
Mechanisms of Coral Bleaching

Coral reefs cover less than 1% of the ocean floor but support more than 25% of all marine life. Estimates from various studies indicate that if current trends continue, such as rising sea temperatures, ocean acidification, pollution, and other stressors,a majority of the world's coral reefs could be lost by 2050. Corals are animals that form a symbiotic relationship with algae, a plant. Corals receive anywhere from 70% to 90% of their energy requirements through photosynthesis performed by algae living within their tissues. Coral bleaching is a phenomenon where coral expel the colorful algae living in their tissues, turning them pale or white. This expulsion is often due to stressors like rising sea temperatures, pollution, or changes in water chemistry. The molecular mechanisms underlying coral bleaching, algae expulsion from coral tissues, remain unclear. Here, we seek to map the biochemical networks in coral and algae that underlie stress sensitivity using mass spectrometry proteomics technology. In collaboration with the Coral Resilience Lab at the University of Hawaii at Manoa, we are quantitatively comparing coral and algae protein expression across various Montipora capitata genotypes that vary in their heat resilience.
Current Projects
Phosphoproteomics of Bleaching in Reef-Building Corals. In the first mass spectrometry phosphoproteomics study ever performed in hard reef-building corals, we study how phosphorylation signaling networks are dysregulated during heat stress and symbiosis breakdown between coral and symbiont, defining stress signaling and other cellular pathways at the host-symbiont interface.
People: Immy A. Ashley, Shaylyn Carthen
Collaborators: Crawford Drury (University of Hawaii), Eva Majerova (University of Hawaii)
Firefighter and Lung Health

Our work, in collaboration with Brigitte Gomperts, Victoria Barber, and David Gonzales at UCLA, and Captain Jeffery Kimura at Fire Station 3 in Los Angeles, integrates chemical exposure science with human airway biology to define how wildfire-related pollutants and occupational contaminants drive airway dysfunction and cancer risk. We use mass spectrometry phosphoproteomics to characterize the impacts of volatile organic compounds (VOCs) and particulate matter that persist in indoor environments and accumulate on firefighter personal protective equipment (PPE) on cellular signaling, specifically primary human airway epithelial air–liquid interface (ALI) models. We have shown that exposure to woodfire smoke activates stress and oncogenic signaling programs, impairs mucociliary clearance, and barrier integrity, establishing a mechanistic link between inhaled byproducts and airway injury. Building on this, we longitudinally profile contaminants that accumulate and evolve on firefighter PPE in Los Angeles with real-world use and assess how degassed PPE-derived chemicals reprogram cellular signaling pathways in airway epithelial cells. By integrating chemical measurements with transcriptomic, proteomic, and phosphoproteomic analyses, and by testing mitigation strategies such as small molecule therapeutics and surface cleaning, our work links persistent smoke and PPE-associated exposures to airway dysfunction and carcinogenic risk while informing evidence-based interventions to reduce long-term respiratory health hazards.
Current Projects
Firefighter Personal Protective Equipment (PPE). In collaboration with Fire Station 3 in Los Angeles, we define the composition and persistence of contaminants that accumulate on firefighter PPE during real-world use and assess their off-gassing and impact on phosphorylation signaling in airway epithelial signaling using primary human ALI models.
People: Declan Winters (T32-CMB Scholar), Immy Ashley, Sara Makanani (HHMI Gilliam Scholar)
Collaborators: Jeffrey Kimura (Fire Station 3); Brigitte Gomperts, Victoria Barber and David Gonzales (UCLA)
Indoor Air Volatile Organic Compounds (VOCs). We characterize VOCs that persist in indoor environments following wildfire exposure and determine their effects on airway epithelial signaling, barrier integrity, and mucociliary function. Using chemical profiling and multi-omics analyses in ALI models, we identify pathways driving airway dysfunction and test interventions to mitigate VOC-induced injury.
People: Declan Winters (T32-CMB Scholar), Immy Ashley, Sara Makanani (HHMI Gilliam Scholar)
Collaborators: Brigitte Gomperts, Victoria Barber and David Gonzales (UCLA)
bottom of page